Abstract
Introduction: Hemophagocytic lymphohistiocytosis (HLH) is a rare, life-threatening hyperinflammatory syndrome characterized by overactivation of the immune system due to systemic release of proinflammatory cytokines, especially of interferon gamma (IFNγ), and persistent activation of macrophages/histiocytes and T cells. Emapalumab, a fully human, anti-IFNγ monoclonal antibody that binds to both free and receptor-bound IFNy, neutralizing its biologic activity, was approved by the Food and Drug Administration (FDA) in 2018 for the treatment of adult and pediatric patients with primary HLH with refractory, recurrent or progressive disease or intolerance with conventional HLH therapy. The safety and efficacy of emapalumab was based on results from a pivotal phase 2/3 trial which reported a 63% overall response in patients treated with emapalumab. Since approval, no study has evaluated the use of emapalumab in a larger cohort of patients in the real-world clinical setting.
Aim: To assess real-world treatment patterns and outcomes, clinical and demographic characteristics among patients treated with emapalumab.
Method: This retrospective, non-interventional, observational medical chart review study will include patients treated across treatment centers in the US with emapalumab in a non-clinical trial setting. The study aims to include more than 100 patients who have been treated with at least one dose of emapalumab between November 20, 2018 and December 31, 2020 (patient identification period). The date that the patient initiates treatment with emapalumab within the patient identification period is defined as the index date. The post-index date is defined as the period from the index date through to the study end date (June 30, 2021), end of data availability for the patient, or date of death, whichever occurs first. Patients will be classified into three groups (i.e. primary HLH, secondary HLH, or non-HLH) based on the information obtained from their charts, the HLH 2004 diagnostic criteria, and adjudication by the REAL-HLH Steering Committee.
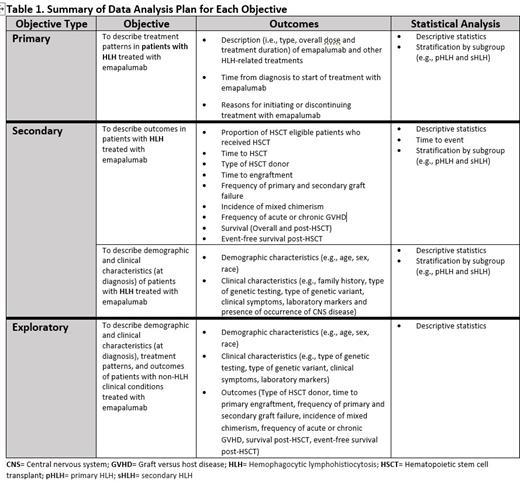
Results: The primary objective of the study is to describe treatment patterns in patients with HLH treated with emapalumab in a real-world clinical setting, including emapalumab dose, treatment duration, and reasons for initiating or discontinuing treatment. The secondary objectives are to describe demographic and clinical characteristics of patients with HLH treated with emapalumab, and their outcomes. The exploratory objectives include detailing the demographic, clinical characteristics, treatment patterns, and outcomes of patients with non-HLH clinical conditions treated with emapalumab (Table 1). Demographics and clinical characteristics will be analyzed at the time of diagnosis. Treatment patterns and outcomes will be analyzed during the post-index date.
Conclusion: The study aims to assess treatment patterns and outcomes, clinical and demographic characteristics among patients treated with emapalumab, a novel IFNγ blocking agent, in real-world clinical settings in the US.
Allen: Sobi: Consultancy. Leiding: Sobi: Consultancy. Chandrakasan: Sobi: Consultancy. Walkovich: X4 Pharmaceuticals: Other: Local PI for clinical trial involving mavorixafor and patients with neutropenia; Swedish Orphan Biovitrum AB (Sobi): Consultancy, Honoraria; Pharming: Honoraria, Membership on an entity's Board of Directors or advisory committees; Horizon Pharmaceuticals: Honoraria, Membership on an entity's Board of Directors or advisory committees. Oladapo: Sobi: Current Employment. Yee: Sobi: Current Employment. Pednekar: Sobi: Consultancy; PRECISIONheor: Current Employment. Raza: Sobi: Consultancy; PRECISIONheor: Current Employment. Jordan: Sobi: Consultancy.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal